
Surrounding the cells in the animal and human body is a fibrous network of proteins and polysaccharides called the extracellular matrix or ECM. This meshwork provides tissues with a scaffold for the structural support needed to build different tissues and maintain their structure.
But the ECM does much more, such as regulating cell growth and differentiation and most remarkably, guiding cells to their right place — their correct zip code — in the body, then keeping them there.
Normal cells cannot survive if they aren’t in contact with the ECM; they undergo apoptosis (see John Reed and the Deathly Revelations of Apoptosis). In cancer, however, this system fails. Cells that have turned malignant acquire the ability to separate from ECM and survive the detachment. These new traits make it possible for cancer cells to leave their original zip code and migrate elsewhere in the body to form metastases, which is what makes cancer so lethal.
In a body of scientific work that spanned almost 20 year, Erkki Ruoslahti, MD, PhD played a leading role in the discovery of ECM adhesion proteins, the cellular receptors that bind the cells to ECM, and the chemistry of the binding interaction.
As a result, we now understand in molecular detail how the body plan is put together during development and how it is maintained through life. Several drugs have been designed and introduced to clinical practice or are in clinical trials based on the work by Ruoslahti and many others on cell adhesion mechanisms.
The first notable milestone in the quest to understand molecular zip codes took place in 1973, when Ruoslahti and Antti Vaheri, assistant professors working at the University of Helsinki in Finland, and Richard O. Hynes working independently at the Imperial Cancer Research Fund in England, discovered a protein that became later known as fibronectin.
Ruoslahti and Hynes later moved to the United States: Hynes as a founding faculty member of the MIT Center for Cancer Research and Ruoslahti to what now is the Sanford Burnham Prebys Medical Discovery Institute.
“When I started my own (laboratory), there was very little known about how cells recognize their surroundings and how they knew where they should go,” Ruoslahti would later recall.
Fibronectin provided answers. It is the prototype adhesion molecule in the ECM, and its responsibilities are myriad. It allows cells to stick to the ECM and sometimes to each other. It guides cell movement during embryonic development and assists in wound repair. It is a key ingredient of blood clots and aids other components in the ECM, such as collagen, heparin and growth factors, to fulfill their roles.
Digging deeper into the biochemistry of fibronectin, Ruoslahti pinpointed the three-amino acid sequence RGD in fibronectin that allows cells to attach to this protein in ECM. The scientific literature now boasts tens of thousands of mentions of the RGD sequence and its roles in biomedical research and health.
With Hynes and Timothy A. Springer, PhD, at Harvard Medical School, Ruoslahti began describing a new class of cell surface adhesion receptors called integrins. Fibronectin is one of the integrin targets in ECM, but there are many others.
“These receptors have so many roles to play and are responsible for an enormous amount of the organization and functioning of the body,” said Hynes in a 2022 interview published in PNAS. “How cells know where to stick and why they don’t always stick and how they let go are important questions in biology and in pathology.”
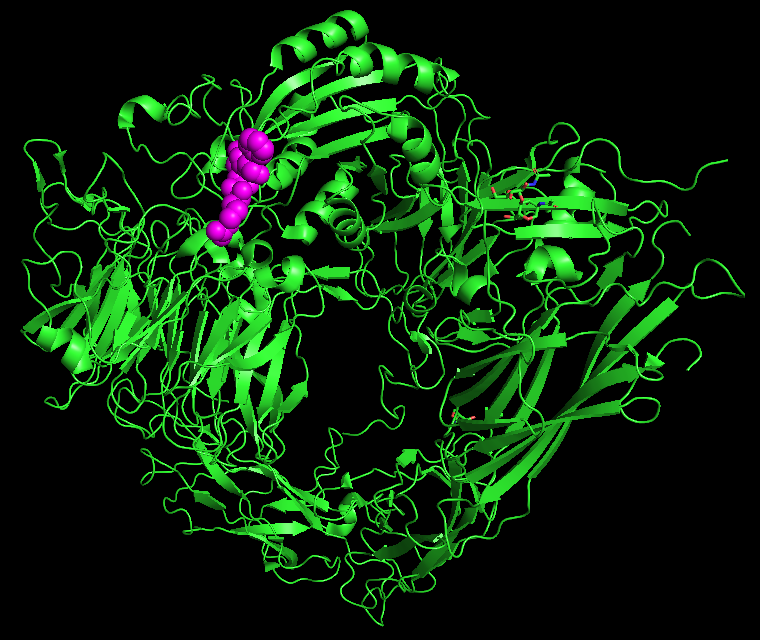
In 2022, Ruoslahti, Hynes and Springer were honored for their combined work with an Albert Lasker Basic Medical Research Award, sometimes referred to as “America’s Nobels.”
The discovery of integrins and subsequent research has profoundly influenced both basic biomedical science and corresponding developments in the treatment of disease. How well integrins do their jobs — or don’t do their jobs — affects a number of critical biological processes, including inflammation, infection and angiogenesis — the development of new blood vessels.
Integrins are pivotal receptors for brain development, function and nerve repair. In cancer, misbehaving integrins can change signaling pathways in cells that allow tumor survival and growth, metastasis and resistance to therapies.
As a result, integrins have become popular targets for therapeutic interventions, including FDA-approved treatments for cardiovascular diseases, inflammatory bowel disease, multiple sclerosis and dry eye disease. Research is ongoing to develop integrin-based therapies for multiple types of cancer.
“One of the integrins is the main receptor in platelets that makes platelets aggregate in blood clotting, and we showed that it also was an RGD receptor,” said Ruoslahti in the 2022 PNAS interview. “Pharmaceutical companies made chemical compounds that mimic the RGD peptide sequence and used them in preventing what is called reocclusion: when an artery gradually closes again after it has been opened up with a balloon. It turned out that reocclusion could be prevented by RGD peptides and RGD-derived compounds.
“Another area where these peptides hopefully will be useful is to target drugs in tumors. Cancer blood vessels are rich in αV integrins, so we developed an RGD peptide that seeks out tumors because it binds to the αV integrins in tumor vessels. It goes deep into tumor tissue, and it can take a payload with it. What’s remarkable is that you don’t have to couple the peptide to the drug, you just give them at the same time, and the endothelial cells take in whatever is there in the blood next to the cell, including the drug, and the drug is then transported throughout the tumor.”
Ruoslahti joined the La Jolla Cancer Research Foundation (precursor to Sanford Burnham Prebys) and served as president and CEO from 1989 to 2002. He continues to work as a Distinguished Professor Emeritus, studying peptides that bind to specific targets in the body, such as tumors, brain injuries, blood vessels in Alzheimer’s disease lesions and wounds.
These peptides can be used to selectively deliver diagnostic probes and drugs to the target. A tumor-homing peptide that improves the penetration and spreading of anticancer drugs in tumors is in advanced clinical trials.
